
Demystifying Sinner's Circle for smart cleaning
Much is said about developing the best product at the lowest cost, but where does development begin? What theoretical foundations can be used to study a new product?
This article will discuss a concept well known to chemists: the "Sinner's Circle" and the different cleaning mechanisms.
It is important to use theory and structure our thinking before moving on to practice.
The concept
Herbert Sinner, head of application technology for detergents at Henkel, coined the concept of Sinner's Circle. According to him, "cleaning is an operation that consists of applying a detergent at a certain temperature for the time necessary to dissolve or soften the residue so that mechanical action can easily remove it!"
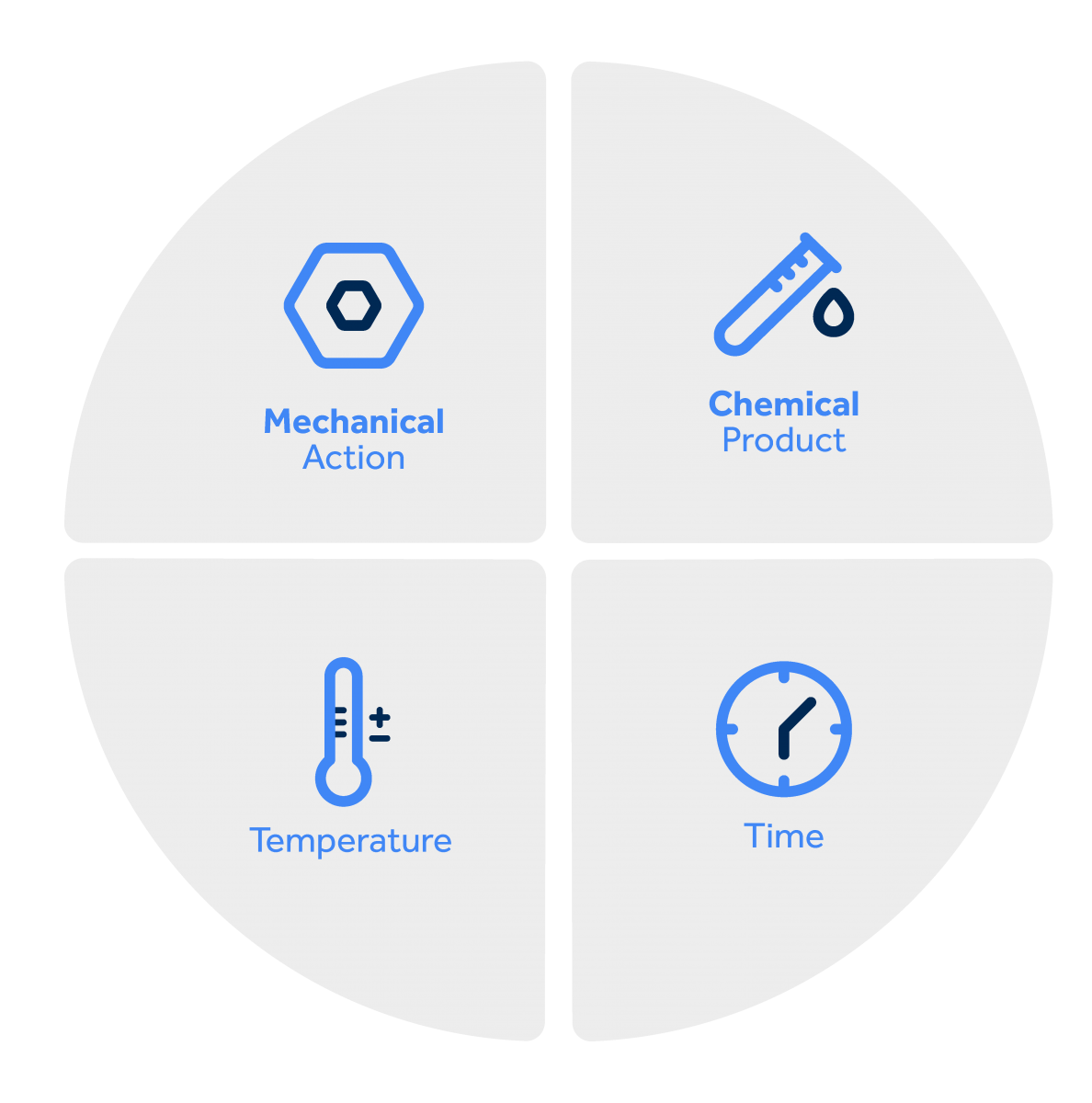
This means that each cleaning method, surface, and type of dirt will require different combinations. Understanding the relationship between these variables is important to achieve the best results.
Now let's take a closer look at each factor to understand how to make the best choices.
Mechanical action
This factor represents the application of physical or mechanical force to remove dirt.
There are some cleaning mechanisms involving mechanical action, such as abrasion, the wearing away of one material by friction with another. An example in our daily lives is when you need to make a pan shine and use steel wool to do so.

Thermal action
The increase in temperature increases the solubility of the medium, enhancing the action of chemicals and consequently assisting in cleaning. In addition, high temperatures can be lethal to many microorganisms, contributing to the disinfection of surfaces.
Heat also denatures proteins present in organic stains such as blood or food, facilitating their removal when washing clothes, for example.
A practical example of where heat is very helpful is in CIP (cleaning in place). The only mechanical action present in this system is the water pressure that circulates internally, which forces us to increase the efficiency of the other points in the Sinner's Circle, compensating for this factor.
The action of time
The exposure time of the chemical product to the surface to be cleaned directly influences its effectiveness. Factors such as the type of dirt and the nature of the surface affect this time, as more porous surfaces require more time to act.
It is important to pay attention to the saturation of the solution, as this reduces its cleaning capacity over time. For this reason, it is important to evaluate the factors to determine the best contact time and concentration required.
A good example of the effect of time is understanding how a disinfectant works.
The action of a disinfectant consists of diffusing into microbial cells and, upon penetrating them, triggering chemical reactions that compromise cellular integrity, leading to the death of the microorganism. This process can occur through protein denaturation, oxidation, or even through alterations in cellular metabolism.
It is also possible to observe that disinfectant labels indicate the minimum contact time required to achieve effectiveness.
Chemical action
This refers to the action of the chemical products used in the process. The study of synergy between the raw materials used to make this product will be fundamental to successful cleaning.
Within the context of chemical reactions, there are some key points to consider, which we will discuss below.
Saponification
Saponification is a chemical reaction that occurs between a triglyceride (fat or oil) and a strong base, such as sodium hydroxide (NaOH) or potassium hydroxide (KOH). During this reaction, the ester bonds of the triglycerides are broken, forming soap molecules (fatty acid salts) and glycerol.
The soap molecule is amphipathic, with one polar end and one nonpolar end. This structure allows soap to bind to both grease and water simultaneously, forming micelles that encapsulate dirt and remove it.
.png)
Surfactants:
Often, the formulation of a cleaning product involves combining different types of surfactants to optimize performance. This synergy can improve dirt removal capacity, foaming, and other desirable properties.
Surfactants are classified according to the nature of the ionic group present in their molecule.
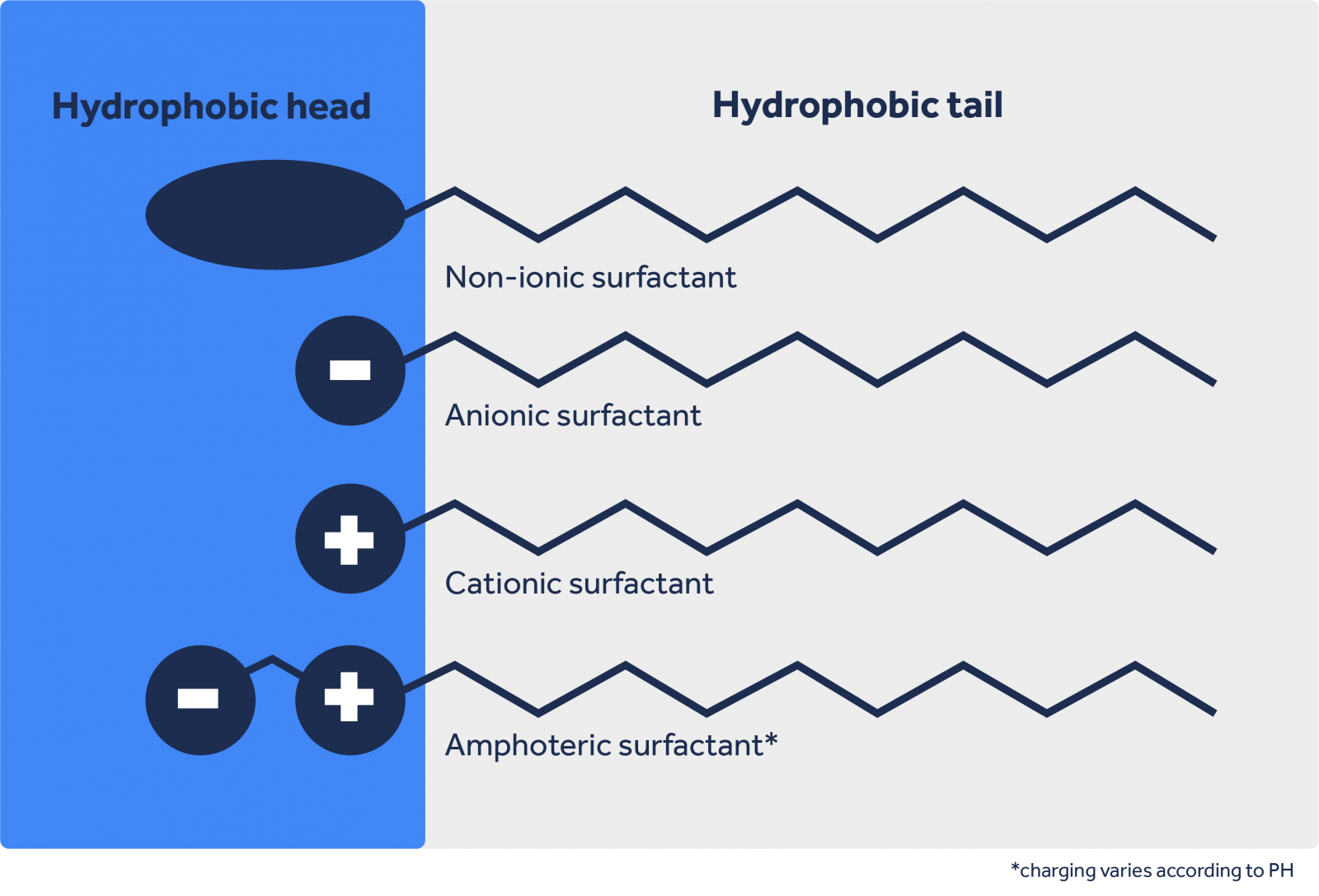
To learn more about the theory behind surfactants, click here.
Emulsion
Emulsion is one of the pillars of the cleaning process. The mechanism starts from the bonds made by the surfactants present in the cleaning products with the dirt on the surface to be cleaned. The emulsion breaks down the dirt into small particles, forming micelles that are dispersed in the liquid and subsequently removed.
These surfactants, called emulsifiers, have a hydrophilic part and a lipophilic part. This characteristic allows them to bind to both water and oil molecules. One way to choose the most suitable surfactant is to use the HLB concept, a scale that indicates the affinity of the substance with water or oil, because the higher the HLB value, the more hydrophilic the surfactant is. On the other hand, the lower the HLB value, the more lipophilic it is.
A practical example of the emulsion acting on the cleansing system is when removing make-up from the face. Emulsifiers bind to make-up and oil particles, enveloping them in small spheres called micelles. These micelles are dispersed in water, making make-up soluble and easy to remove. When you rinse your face, the micelles, along with the make-up and excess oil, are removed from the skin.

Solvency
An old Brazilian saying, often heard during school days, perfectly defines this cleaning mechanism: “similar dissolves similar”. This sentence summarizes a solvent as a substance capable of dissolving other substances, called solutes.
The ability of the solvent depends on its chemical nature; for example, water is an excellent solvent for polar substances, such as salt, but does not dissolve nonpolar substances, such as fat, well.
In turn, organic solvents such as acetone and benzene dissolve nonpolar substances well, such as oils and greases, and are used in specific cleaning products, such as paint and enamel removers.

Oxidation-reduction
These are complementary physicochemical processes: oxidation and reduction. In oxidation, a substance loses electrons during a chemical reaction, while in reduction, a substance gains electrons during a reaction. They occur simultaneously; while the reducing agent is a substance that donates electrons and undergoes oxidation, the oxidizing agent is a substance that accepts electrons and undergoes reduction.

A good example of this process is the action of hydrogen peroxide, which performs an oxidizing action. It bleaches fabrics because when it comes into contact with dye molecules, these undergo oxidation. This oxidation breaks the chemical bonds that give color to the fabric, fragmenting the dye molecules into smaller, colorless molecules.
In addition to whitening fabric, hydrogen peroxide is also effective in removing stains, such as blood, wine, and coffee. It oxidizes the substances that cause these stains, making them more soluble and easier to remove.
Pickling
Pickling is a chemical process that uses acids to remove impurities and oxides from the surface of metals. The choice of acid, the concentration of the solution, and the process conditions are crucial factors in ensuring the efficiency and safety of the process.
Acid pickling, the most common method, is based on the reaction between the acid and the metallic oxides present on the surface. The acid, usually hydrochloric (HCl) or sulfuric (HSO), reacts with the oxides, forming water-soluble salts and releasing hydrogen.
Example with Hydrochloric Acid and Iron Oxide:
Considering iron(III) oxide (FeO), one of the most common impurities on metallic surfaces, the reaction can be represented as follows:
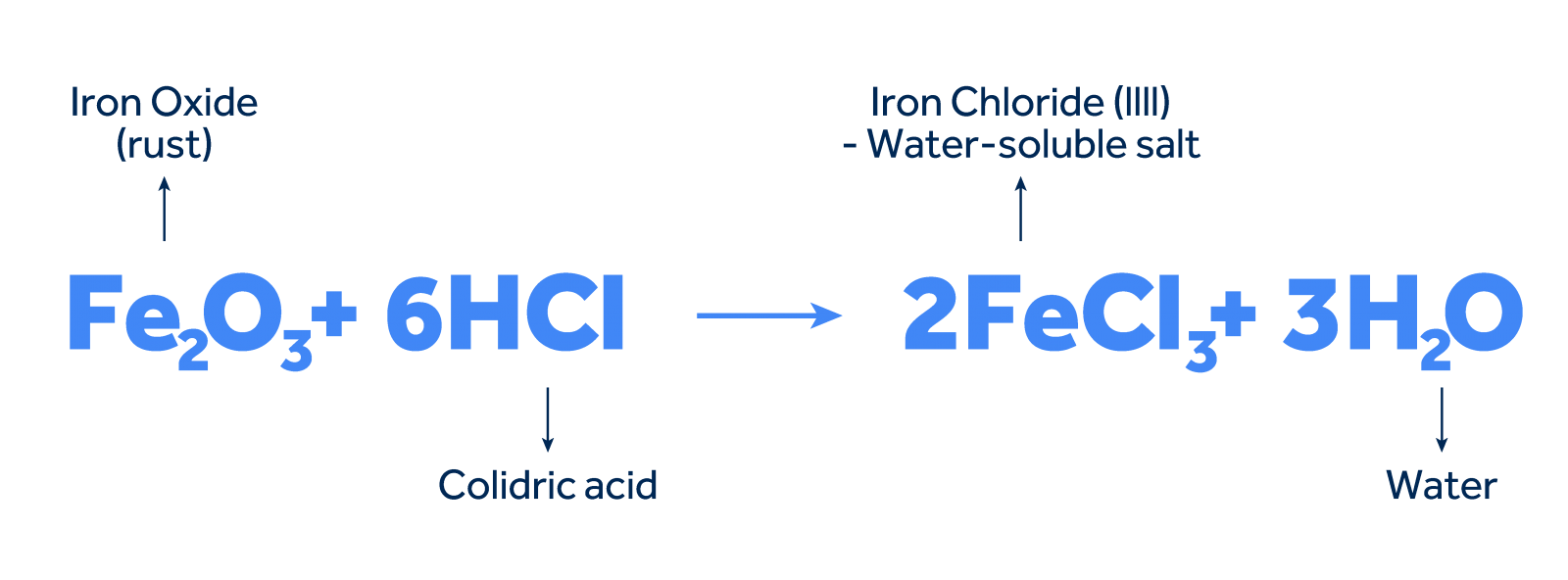
What happens in the reaction:
- Dissolution of the oxide: Hydrochloric acid "attacks" the iron oxide layer, breaking chemical bonds and forming iron ions and chloride ions.
- Salt formation: Iron and chloride ions combine to form iron(III) chloride, a water-soluble salt.
- Hydrogen release: The reaction also releases hydrogen gas, which is released from the solution.
The iron(III) chloride formed in the reaction is soluble in water and easily removed from the metal surface by rinsing.
The factors that influence pickling and will guide the choice of the best acid and process will depend on the type of metal, the thickness of the oxide layer, temperature, agitation, and other components that can influence this system, such as paint on the parts or dirt from oils and greases.
The importance of the Sinner's Circle
By demystifying Sinner's Circle, we realize the complexity of cleaning and the importance of each step. In short, it guides us towards efficient cleaning, but it is the personalization of each step that guarantees success.
Macler, with its experience and technical expertise, offers customized solutions for each challenge, ensuring the selection of the most suitable products and the optimization of each stage of the process.
Count on Macler as a chemical and strategic partner to find the ideal combination for your business needs!

Learn More
Perhaps one of the best-known chemicals used by Brazilian consumers, which has a dramatic influence on culture and behavior with regard to cleaning, is sodium hypochlorite, commonly known as “bleach.”
Our chemistry
We use our labs to create
intelligent chemical solutions balanced with your reality.
Products
We use our R&D center, our own laboratory with experienced professionals, to deliver intelligent chemical solutions in balance with your reality.
