
Nonylphenol Ethoxylate vs. Isogen E-TEC: a toxicity comparison
Nonylphenol Ethoxylates are a class of non-ionic surfactants widely used in cleaning products for over 60 years. Their characteristics, such as good detergency, emulsifying capacity, and excellent cost-effectiveness, have made them a constant presence in various consumer items.
However, with increased awareness of the environmental impacts of chemical substances, the use of Nonylphenol Ethoxylate has begun to be questioned. Its presence in the environment, even in extremely low concentrations, can lead to serious ecological and public health problems.
In response to this environmental concern, Macler developed the Isogen E-TEC, an alternative to Nonylphenol Ethoxylate, formulated to replace traditional surfactants in various applications. Isogen E-TEC was designed to offer the same functional qualities, such as emulsifiability, while reducing toxicity and environmental impact.
Continue reading to see a comparison of the toxicity of Nonylphenol Ethoxylate with that of Isogen E-TEC, taking into account various aspects of environmental safety and health effects.
Toxicity: Nonylphenol Ethoxylate vs. Isogen E-TEC
The table below summarizes the acute and chronic toxicity characteristics of Isogen E-TEC and Nonylphenol Ethoxylate between 4 and 15 ethoxylations:
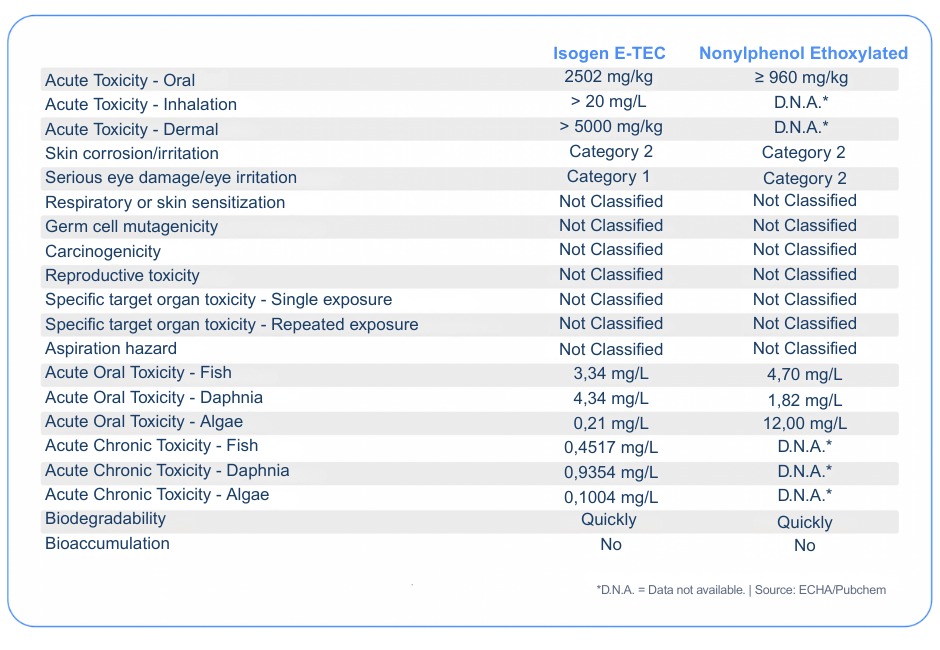 When evaluating this table at first glance, it is already noticeable how little toxicity information is available for Nonylphenol Ethoxylate. However, even so, it is possible to note a certain similarity in various levels of toxicity and risks involving both substances. By analyzing only the data required in a SDS (Safety Data Sheet), we can easily be led to believe that Nonylphenol Ethoxylate is less toxic than Isogen E-TEC.
When evaluating this table at first glance, it is already noticeable how little toxicity information is available for Nonylphenol Ethoxylate. However, even so, it is possible to note a certain similarity in various levels of toxicity and risks involving both substances. By analyzing only the data required in a SDS (Safety Data Sheet), we can easily be led to believe that Nonylphenol Ethoxylate is less toxic than Isogen E-TEC.
Biodegradability
But is that initial impression true? The answer is: no. Nonylphenol Ethoxylates are considerably more toxic than Isogen E-TEC. And why? The key to this question lies within the context of biodegradability.
Nonylphenol ethoxylates degrade rapidly in the environment, with approximately 81% biodegradation in 28 days. However, the main problem lies in the substances generated during this biodegradation, especially nonylphenol, which is extremely toxic and has a high potential for bioaccumulation. Nonylphenol is released rapidly after the biodegradability of Nonylphenol Ethoxylates, and its presence in the environment can be disastrous.
The table below compares the toxicity characteristics of Isogen E-TEC containing nonylphenol, the toxic byproduct of the biodegradability of nonylphenol ethoxylates:
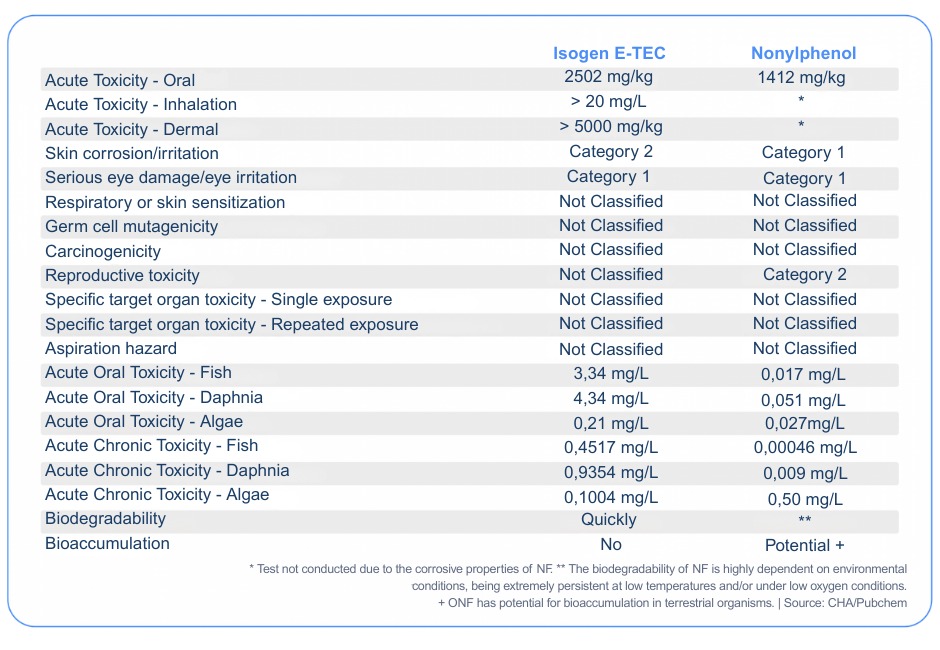 The data demonstrate that Nonylphenol is significantly more toxic than Isogen E-TEC, especially when considering its impact on aquatic life. Nonylphenol has much higher toxicity at extremely low concentrations, which can cause serious damage to aquatic ecosystems. Furthermore, it has the potential for bioaccumulation, meaning it can concentrate in living organisms over time, exacerbating its negative effects.
The data demonstrate that Nonylphenol is significantly more toxic than Isogen E-TEC, especially when considering its impact on aquatic life. Nonylphenol has much higher toxicity at extremely low concentrations, which can cause serious damage to aquatic ecosystems. Furthermore, it has the potential for bioaccumulation, meaning it can concentrate in living organisms over time, exacerbating its negative effects.
Environmental Effects of Nonylphenol
In addition to toxicity information, nonylphenol has a considerable environmental impact, with several harmful effects on different forms of life:
- Endocrine Disruptor: It is classified as a potential endocrine disruptor, affecting the hormonal system of humans and animals.
- Effects on Aquatic Life: At concentrations as low as 8.2 µg/L, it can cause feminization of aquatic organisms, affecting fertility and offspring survival rates.
- Damage to Mollusks: Studies by organizations such as Embrapa and Fiocruz indicate that nonylphenol and its derivatives significantly affect the reproduction and embryonic development of mollusks at concentrations much lower than the levels indicated in toxicity tables.
- Impacts on Nematodes: Both nonylphenol ethoxylates and nonylphenol cause damage to nematode growth and locomotion, although ethoxylates are slightly less toxic than nonylphenol.
Risk assessment at use concentrations
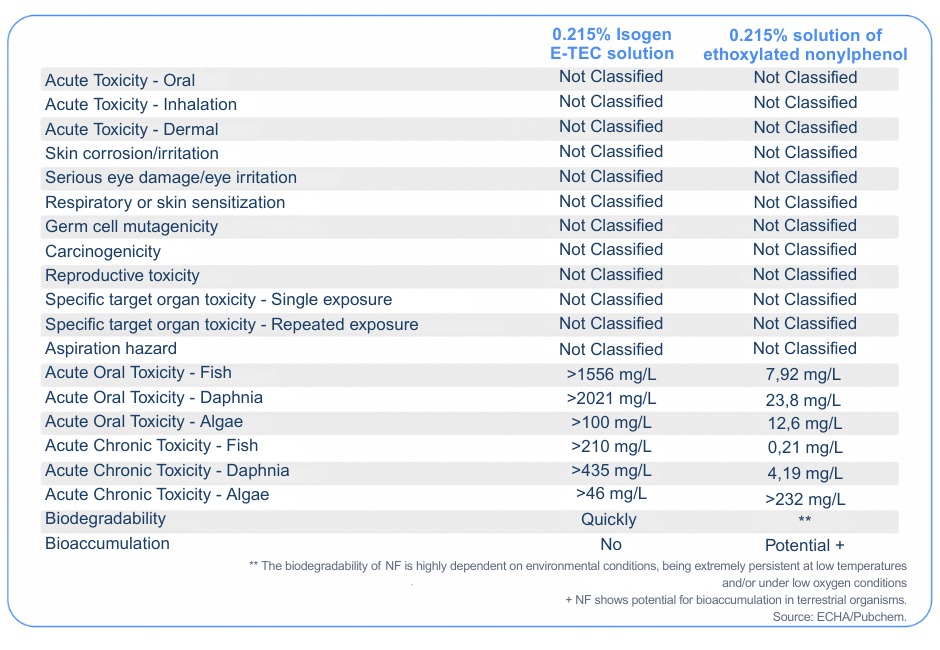
Although the toxicity of the compounds in their pure form is a concern, it is important to consider that surfactants will not be used in their concentrated form. ABNT NBR 14725/23 suggests that Isogen E-TEC, when used in concentrations below 0.215%, does not pose significant risks. However, the nonylphenol residue generated by the biodegradability of nonylphenol ethoxylates may continue to pose risks even at low concentrations.
The following table illustrates the environmental and toxicological risks of diluted solutions of Isogen E-TEC(0.215%) and Nonilfenol (0.215%) under the same conditions:
 Isogen E-TEC: a promising alternative
Isogen E-TEC: a promising alternative
The biodegradability data and environmental impact of nonylphenol reveal that ethoxylated nonylphenol can cause much more serious damage to the environment. The rapid release of nonylphenol after biodegradation makes the environmental effects even more worrying, with the risk of bioaccumulation and disruptive effects on aquatic ecosystems and terrestrial organisms.
(3).png) In summary, replacing Nonylphenol Ethoxylate with Isogen E-TEC not only brings benefits in terms of safety and functionality but also represents a significant step towards reducing the environmental impact of cleaning products and other industrial applications.
In summary, replacing Nonylphenol Ethoxylate with Isogen E-TEC not only brings benefits in terms of safety and functionality but also represents a significant step towards reducing the environmental impact of cleaning products and other industrial applications.
Contact Macler and replace Nonylphenol Ethoxylate in your formulations. Click here.

Learn More
Although practical, traditional cold fabric softeners lose out in perfume fixation and can stain clothing. To resolve this, Macler created a base that unites the savings of this process with high sensory performance. Get to know it.
Our chemistry
We use our labs to create
intelligent chemical solutions balanced with your reality.
Products
We use our R&D center, our own laboratory with experienced professionals, to deliver intelligent chemical solutions in balance with your reality.