
Sequestrants: function and importance in formulations
When discussing sanitizing product formulations, it is common to focus on the main active ingredients. However, in addition to these, some raw materials are of paramount importance and significantly affect formulations, such as sequestering agents, which we will discuss in this article.
What are sequestering agents?
Sequestrants, also known as chelating agents, are substances whose molecules have a very distinct characteristic, as their molecular structure presents a peculiarity: they have a structure with regions that typically resemble teeth or claws.
This distinctive structure is also responsible for the name chelating agent, derived from the Greek word chele, which means claw.
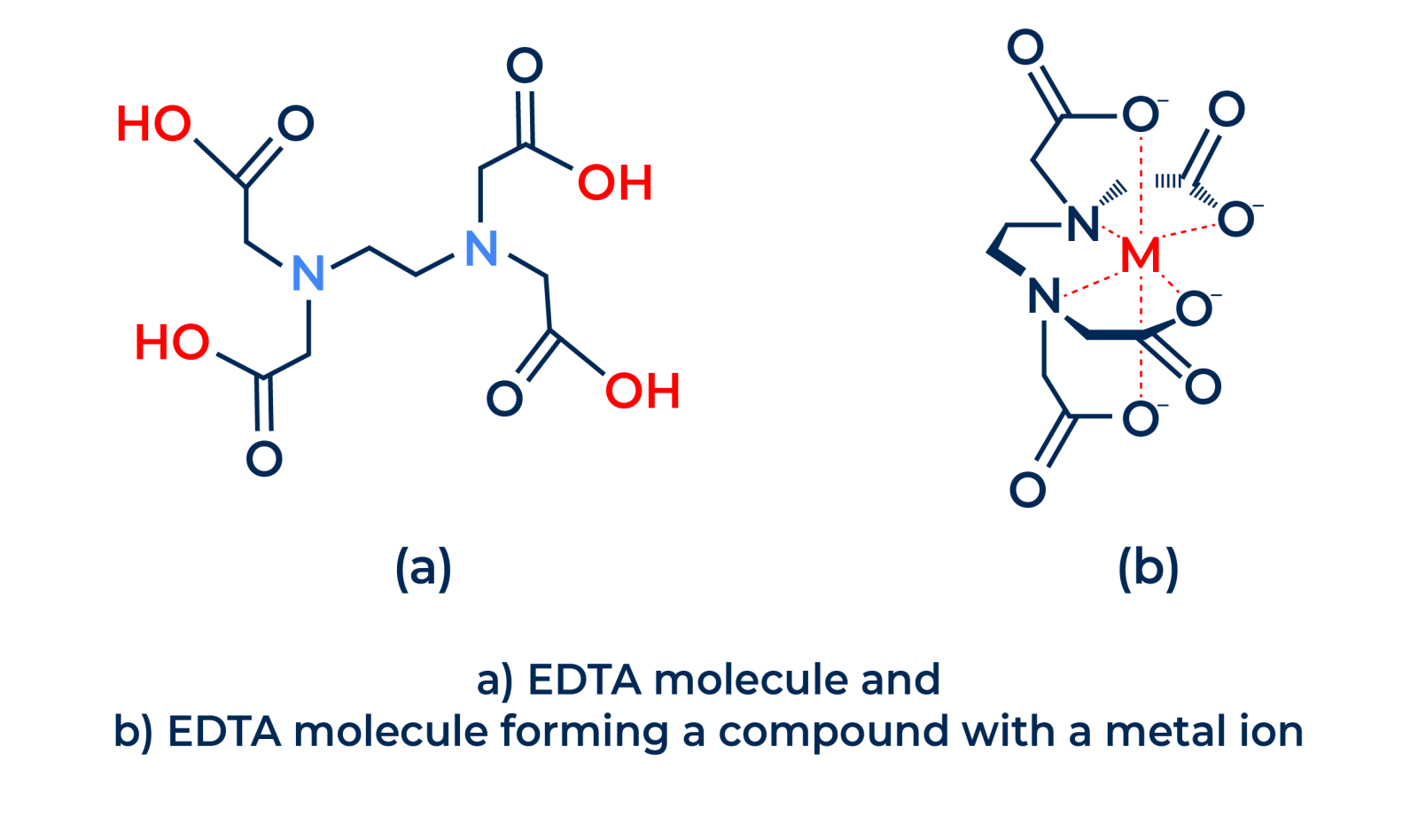
Let's visualize these claws in the structure of a famous sequestrant, EDTA. Below, we have the structure of this sequestrant molecule, with its claws highlighted in red:
.png)
These claws are not only visually striking, but also possess a unique ability: they can "pinch" or "trap" metallic ions, removing them from the aqueous environment through a process known as complexation.
From a more technical perspective, sequestering agents are molecules responsible for forming a ring-shaped complex through conjugation with metal ions via dative covalent bonds; in other words, they are molecules responsible for forming complexes with metal ions, sequestering them.
This ability to "trap" metal ions prevents them from interfering with other components of the formula, ensuring the stability, effectiveness, and safety of the final product.
Therefore, in a sanitizing formulation, sequestering agents end up being responsible for protecting the active ingredient, since potable water will always contain a certain amount of dissolved metal ions, which is also known as water hardness.
In formulations with cationic surfactants, sequestering agents protect the active ingredient by sequestering ions from water hardness, increasing the effectiveness in removing cations that are important and necessary for the membrane activity of microorganisms.
In formulations with anionic surfactants, sequestering agents enhance the performance of the active ingredient and improve attributes such as foaming.
How do sequestrants protect the cationic asset?
Cationic compounds, present in disinfectant formulations, for example, are protected by sequestrants in different ways:
- Complexation of metal ions: Ion scavengers bind to metal ions present in water, primarily calcium and magnesium, forming stable complexes. This prevents the metal ions from binding to the cationic active ingredient of the disinfectant, reducing its effectiveness. This reduced effectiveness is because the cationic active ingredient ends up "confusing" the calcium and magnesium ions with the membrane of microorganisms and "attacking" these ions instead of fulfilling its role of attacking and eliminating the microorganism.
- Precipitation prevention: Metal ions can react with the cationic active ingredient in disinfectants and form insoluble precipitates. This can reduce the concentration of the cationic active ingredient in the solution and make the disinfectant less effective. Ion scavengers bind to metal ions and prevent them from reacting with the cationic active ingredient, thus preventing the formation of precipitates.
- Improved stability: Ion scavengers can help protect the cationic active ingredient from degradation by oxidation or other chemical reactions. This can improve the disinfectant's stability and increase its shelf life. They prevent oxidation reactions of metals from occurring in dyes and fragrances.
How do sequesters protect anionic assets?
The use of sequestering agents in regions with high water hardness is essential to guarantee product quality and stability. The sequestering agent prevents calcium and magnesium ions from reacting with the detergent's surfactants, protecting them and ensuring foaming and product stability, since hard water directly impacts foaming due to the presence of metallic ions.
These ions react with the surfactants in the detergent, which are responsible for foam formation, and form insoluble precipitates. This reduces the amount of surfactants available to form foam, resulting in less abundant and finer foam.
Although the amount of foam is not necessarily an indicator of detergent effectiveness, it is directly associated with poor quality by the consumer; furthermore, if the active ingredient is insoluble, it will not have cleaning capacity.
Examples of sequestrants
Each sequestrant has specific characteristics and applications, making it essential to choose the ideal one for each need.
EDTA (Ethylenediaminetetraacetic Acid)
As previously mentioned, EDTA is one of the most widely used chelating agents due to its high efficiency and versatility, being able to bind to a wide range of metal ions. However, it is also a very selective chelating agent, meaning it can bind to specific metal ions without binding to other ions present in the solution.
It is effective in complexing a wide range of metal ions, including Calcium, Magnesium, Iron, and Copper. It is relatively inexpensive and easy to use.

EDTA sequesters metal ions through a process known as chelation. The EDTA molecule has six electron-donating groups: four carboxylic groups and two amine groups. These electron-donating groups bind to metal ions, forming a stable complex. The strength of the bond between EDTA and the metal ion depends on the charge of the metal ion, the size of the metal ion, and the nature of the electron-donating groups in EDTA.
EDTA performs a chelating process, "embracing" the metal ion and protecting the other components of the formulation.
Although it is a widely used sequestrant, there are currently some points that should be considered when opting for the use of EDTA. Firstly, regarding environmental impacts, EDTA is not a biodegradable substance, a fact that can generate serious problems because its presence in the environment can impoverish the soil and remove substances necessary for life in both soil and water.
The risks posed by this substance should also not be overlooked: in its solid form, EDTA is a powder that can cause serious eye damage, as well as being a strong respiratory irritant and harmful if swallowed. Its diluted form eliminates the risk of respiratory irritation, but the other problems remain.
This set of characteristics generated a driving force for the development and use of alternative sequestrants to EDTA, especially aimed at combating the environmental damage that this substance is capable of causing.
NTA (Nitrilotriacetic acid)
Nitrilotriacetic acid is a more environmentally viable option than EDTA, as it is a biodegradable substance. This sequestrant is obtained as a byproduct of EDTA synthesis, but it can also be manufactured through chemical synthesis. However, this synthesis is not very simple, since it requires extremely toxic reagents such as formaldehyde, sodium cyanide, and hydrogen cyanide.
In addition to the toxicity present in the synthesis of this product, it is worth highlighting that this sequestrant has a higher toxicity than EDTA. In other words, even though it is a biodegradable alternative sequestrant, its use implies several risks, among which are irritation to the eyes, skin, and respiratory tract, as well as being classified as a possible carcinogen for humans.
Therefore, although this sequestrant is effective in complexing metal ions such as calcium, magnesium, and iron, its use should be very carefully considered due to the inherent risks of the substance.

DTPA (Diethylenetriaminepentacetic acid)
DTPA is a more selective and effective sequestrant in complexing some metal ions than EDTA. However, it should be noted that this sequestrant has very low solubility in water, unlike its competitor, EDTA.
DTPA is an octadentate ligand, which gives this substance exceptional chelating capacity. With this characteristic, this sequestrant is able to form complexes even with radioactive elements such as thorium, uranium, and plutonium. These complexes facilitate the safer excretion of these substances through urine, making the medicinal potential of this sequestrant interesting. In addition to its use in radiation treatment, DTPA can also be used as a contrast agent for magnetic resonance imaging (MRI) scans.
Given this, DTPA is an extremely potent chelating agent, but its uses in the pharmaceutical and medical fields make its cost typically not viable for most applications in the chemical industry.

HEDP (1-Hydroxy-Ethylidene-1,1-Diphosphonic Acid)
1-Hydroxyethylene 1,1-diphosphonic acid, or etidronic acid, or simply HEDP, is another common sequestrant alternative available on the market. A key difference between this sequestrant and the others presented here is that HEDP is not a nitrogenous molecule, but rather a bisphosphonate.
Regarding environmental impacts, HEDP presents a similar problem to EDTA: it is also not a biodegradable molecule. However, this lack of biodegradation can generate even greater impacts in the case of HEDP, as this molecule is strongly adsorbed by the soil, which considerably increases phosphorus levels in the soil.
Phosphorus is an element that causes considerable concern, since its accumulation in the environment generates significant damage. The presence of this element can even be prohibitive in numerous products, as the sale of products containing phosphorus is highly restricted in several countries.
This sequestrant exhibits a better affinity for the Fe³ anion, but, in general, its performance is similar to that of EDTA. It should also be noted that HEDP is less stable in chlorine-containing media than EDTA, which could be a problem in applications involving products containing sodium chloride.

GLDA (Tetrasodium Glutamic Acid Diacetate)
GLDA is a powerful and readily biodegradable green sequestrant produced from a natural amino acid. Beyond its strong appeal of low environmental impact, GLDA also stands out as a sequestrant with very low toxicity: compared to EDTA, which presents risks if ingested, can cause serious eye damage, and is a strong respiratory irritant, GLDA does not exhibit any type of acute toxicity and is not considered an irritant, thus enhancing the range of applications for this substance.
Now, let's look at what other benefits GLDA offers compared to other sequestrants, besides its biodegradability. One immediately noteworthy and very interesting property of GLDA is its effectiveness in combating both Gram-negative and Gram-positive molds and bacteria, as it can deprive cell walls of cations, weakening them and enhancing the action of other biocidal substances. Several tests have indicated that adding GLDA to biocidal formulations reduces formulation costs by around 30%, without affecting performance.
Finally, we can evaluate and compare the chelation equivalent of GLDA and EDTA. The chelation equivalent is the amount of pure substance, i.e., with 100% active ingredient, that is needed to remove 1 g of some type of metal from the solution. The following data is valid for a pH range of 7 to 10.

Note that the chelation equivalents of the two products are very similar, despite a slight advantage for GLDA. That is, as the data in this table indicate, it is possible to replace EDTA with GLDA in a 1:1 ratio.
In summary, we can see that GLDA proves to be an excellent substitute for EDTA, offering a less environmentally damaging alternative with lower toxicity and equivalent performance.

How to choose the ideal sequestrant?
Choosing the ideal sequestrant for a formulation is a strategic decision that must take several factors into account.
Among the most important are:
- The metallic ions present: It is essential to identify which metal ions are present in the water or other ingredients of the formulation, as each sequestering agent has an affinity for certain types of ions.
- The desired properties of the product: The sequestering agent must be compatible with the desired properties of the final product, such as viscosity, texture, color, scent, and stability.
- The cost-benefit ratio: The cost of the sequestration should be considered in conjunction with its benefits to the end product.
Choosing the right sequestrant ensures the quality and safety of the final product, as well as contributing to the optimization of its costs and performance.
For guidance in choosing the ideal sequestrant for each application, contact our SmartLab.

Learn More
Our chemistry
We use our labs to create
intelligent chemical solutions balanced with your reality.
Products
We use our R&D center, our own laboratory with experienced professionals, to deliver intelligent chemical solutions in balance with your reality.